
Additionally, baryons such as protons and neutrons ( subatomic particles composed from three quarks) and some atoms (such as helium-3) are fermions, and are therefore described by the Pauli exclusion principle as well. Fermions include elementary particles such as quarks, electrons and neutrinos. The Pauli exclusion principle describes the behavior of all fermions (particles with half-integer spin), while bosons (particles with integer spin) are subject to other principles. This reasoning does not apply to bosons because the sign does not change. However, the only way a total wave function can both change sign (required for fermions), and also remain unchanged is that such a function must be zero everywhere, which means such a state cannot exist. So, if hypothetically two fermions were in the same state-for example, in the same atom in the same orbital with the same spin-then interchanging them would change nothing and the total wave function would be unchanged. This means that if the space and spin coordinates of two identical particles are interchanged, then the total wave function changes sign for fermions, but does not change sign for bosons. Any number of identical bosons can occupy the same quantum state, such as photons produced by a laser, or atoms found in a Bose–Einstein condensate.Ī more rigorous statement is: concerning the exchange interaction of two identical particles, the total (many-particle) wave function is antisymmetric for fermions and symmetric for bosons. Particles with an integer spin ( bosons) are not subject to the Pauli exclusion principle. However, the two values of the m s (spin) pair must be different, so these two electrons will present opposite half-integer spin projections, namely 1/2 and −1/2. Thus, if two electrons reside in the same orbital, then the two values of the pairs, respectively, for the n, ℓ, and m ℓ numbers will be the same. In the case of electrons in atoms, the exclusion principle can be stated as follows: in a poly-electron atom it is impossible for any two electrons to have the same two values for each pair, respectively, at all the four-member set of their quantum numbers, which are: n, the principal quantum number ℓ, the azimuthal quantum number m ℓ, the magnetic quantum number and m s, the spin quantum number. This principle was formulated by Austrian physicist Wolfgang Pauli in 1925 for electrons, and later extended to all fermions with his spin–statistics theorem of 1940. 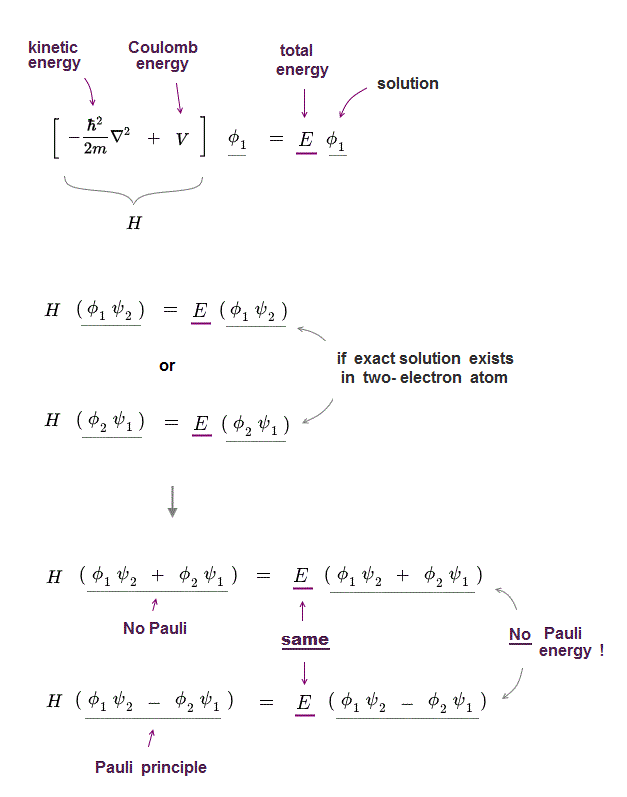 
fermions) cannot simultaneously occupy the same quantum state within a quantum system. In quantum mechanics, the Pauli exclusion principle states that two or more identical particles with half-integer spins (i.e.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
